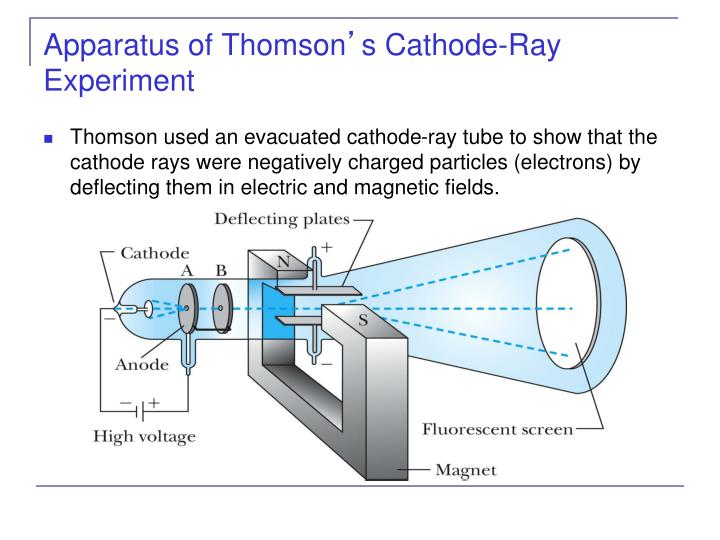
He won the 1906 Nobel prize for the discovery of electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson, a British physicist, conducted the cathode ray experiment. Change of ratio of $e/m$ cathode ray particles are independent of changes in gas which is plaid in the discharged tube.Previously, atoms were known to be indivisible, but in 1897, J. Therefore, the statement given in option (B) is wrong.
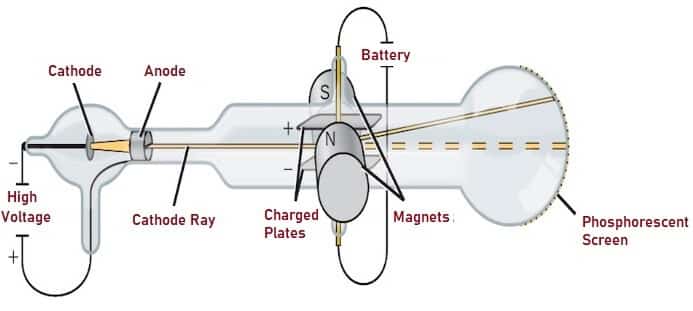
Also he couldn’t show that all the mass contained in atoms is because of the mass of the nucleus only. Note: Thomson could not explain the stability of structure of atom and atomic spectra. Therefore, option (A) and (C) are correct options. In this experiment we had an tube at one end of it, high potential has attached because of which $$ is much greater than $e/m$ of protons. phosphorescence is caused by cathode rays and at one time there was a keen. The number of electrons in an atom are such that the atom, as a whole, has zero charge.įrom the above statement we can say that emission of negatively charged particles from elements indicated that atoms could be split into positive and negative charged particles and atoms possess some internal structure. electricity enters the tube - can be shown in the following way (the experi. According to him, an atom consists of a positively charged sphere with negatively charged electrons embedded in it. Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny dense positively-charged nucleus. Thomson, is one of the most well-known physical experiments, which led to the discovery. Thomson proposed his atomic model in 1903. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. Abstract: The Cathode Ray Tube (CRT) experiment performed by J. As scientists began to examine atoms, their first discovery was that they could extract negatively charged. Later it was believed that atoms were invisible until J.J Thomson discovered the electron in 1897. VCL 2-1: Thomson Cathode Ray Tube Experiment. This experiment also provided an estimate of the ratio of the charge. Thomson was only able to prove the existence of the negatively charged electron. So much so, that George Stoney (1891) proposed the name electron for the particle. From the cathode ray tube experiment, J.J. In some experiments, two electrically charged plates and a magnet were added to the outside of the cathode ray tube. Scientist had believed in the existence of a negative particle for some time. Thomson demonstrated the existence of the electron. They were first observed as experiments in gas discharge tubes started to exploit better and better vacuums (the early experiments observed the varying forms of discharge in low pressure gases cathode rays only become significant when there is very little gas left in the tubes). By balancing the effect of a magnetic field on a cathode-ray beam with an electric field, Thomson was able to show that cathode 'rays' are actually composed of particles. The cathode ray tube experiment performed by J.J. Thomson, (Received October, 1897) The experiments1 discussed in this paper were undertaken in the hope of gaining, some information is to the nature of the Cathode Rays. Cathode 'rays' had been known for some time before Thomson. In some experiments, however, I placed inside the tube a screen, near to the slit through which the cathode rays came, and found that no appreciable. Dalton said that atoms combine to form a molecule which is the smallest unit of a compound. Thomson found that the cathode rays can be deflected by an electric field, as shown below. Elements in atoms are identical and have a characteristic mass. According to him all matter on the earth is composed of small, hard, invisible particles called atoms. As we all know beforehand, the theory of J.J Thomson’s on atom, the john Dalton proposed his atomic theory in 1808.
